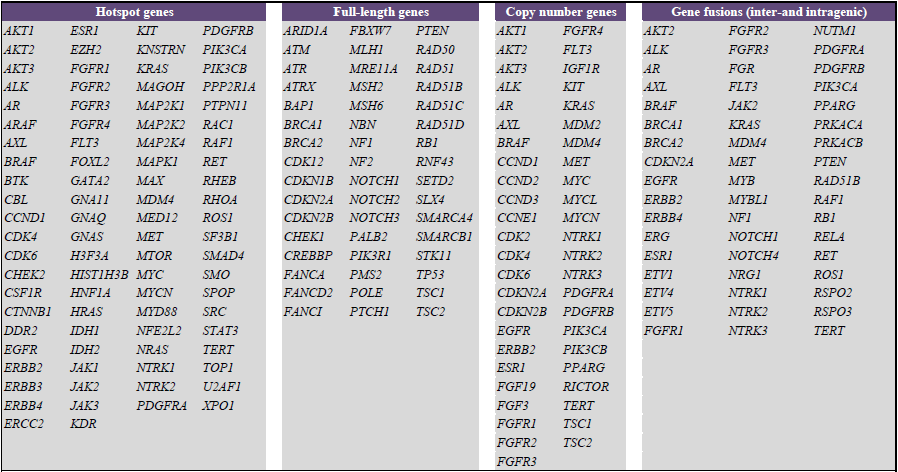
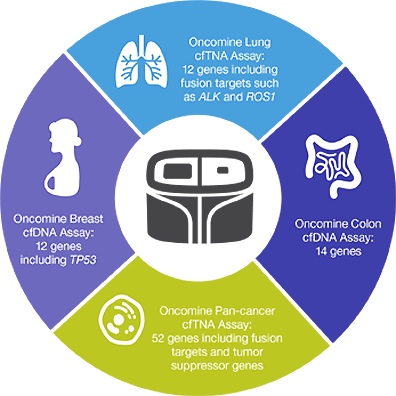
Thermo Fisher Scientific on LinkedIn: Thermo Fisher Scientific Announces Oncomine Clinical Research Grant…

The U.S FDA designates Thermo Fisher Scientific's Oncomine Precision Assay a breakthrough device, ET HealthWorld

FDA Approves First NGS-Based Companion Diagnostic for RET Fusion-Positive Non-Small Cell Lung Cancer

Thermo Fisher and Oncocyte to Jointly Develop IVD Test Kits and Companion Diagnostic Tests in Precision Oncology - Industry News - mobile.Labmedica.com

Perspective: Thermo Fisher Aims to Reduce Time-to-Results for Cancer NGS | Precision Medicine Online

Thermo Fisher on X: "1 test. 1 sample. 1 report. Oncomine Dx Target Test. Offer your oncologists rapid, actionable results. https://t.co/tI76Mip9Nh https://t.co/6ZjFxNdmg6" / X

Thermo Fisher's Oncomine Dx Target Test Receives the US FDA's Approval as a CDx for the Treatment of IDH1-Mutated Cholangiocarcinoma

Thermo Fisher Scientific Announces FDA Approval of Oncomine Dx Target Test as the First NGS-Based Companion Diagnostic to Aid in Therapy Selection for Patients with RET Mutations/Fusions in Thyroid Cancers | Business

Thermo Fisher Scientific Announces Oncomine Clinical Research Grant Awardees and Opens Spring 2023 Submissions